Hess' Law
At AS Unit 2 we used thermodynamic cycles whilst using Hess' law on enthalpy change.
At A2 Unit 5 we use Born-Haber cycles.
At A2 Unit 5 we use Born-Haber cycles.
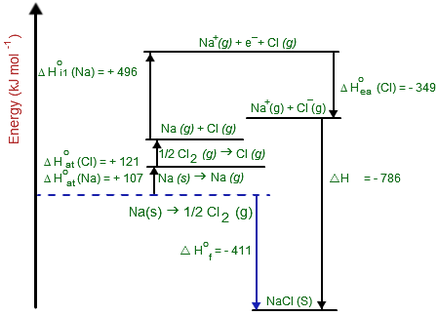
The above image shows the Born-Haber cycle for the reaction Na(s) + 1/2Cl2(g) → NaCl(s)
This shows that the enthalpy of formation (blue arrow) for this reaction is equivalent to:
- The atomisation enthalpy for Na and Cl (Beware about diatomic elements). This is the enthalpy change to get the elements into gaseous state
- The first ionisation enthalpy for Na (In group 2 elements you will need the second ionisation enthalpy)
- The electron affinity enthalpy for Cl
- The Lattice association enthalpy for NaCl from its gaseous ions
When working with the born-haber cycle you can work out questions about working out an enthalpy in the reaction there are different ways you can solve this sort of question.
Mathematical equation:
ΔHform(NaCl) = ΔHat(Na) + ΔHat(1/2Cl2) + ΔH1st ion(Na) + ΔHea(Cl) + ΔHLa(NaCl) (or - ΔHLattice dissociation(NaCl))
Rearrange for the enthalpy you wish to find
Modulus Method:
The Modulus of a number is its magnitude or value. This ignores any sign put in front of it
So the modulus of -5 is 5.
This can be applied when using Born-Haber cycles.
Both sides of the cycle should have the same modulus value.
Mathematical equation:
ΔHform(NaCl) = ΔHat(Na) + ΔHat(1/2Cl2) + ΔH1st ion(Na) + ΔHea(Cl) + ΔHLa(NaCl) (or - ΔHLattice dissociation(NaCl))
Rearrange for the enthalpy you wish to find
Modulus Method:
The Modulus of a number is its magnitude or value. This ignores any sign put in front of it
So the modulus of -5 is 5.
This can be applied when using Born-Haber cycles.
Both sides of the cycle should have the same modulus value.
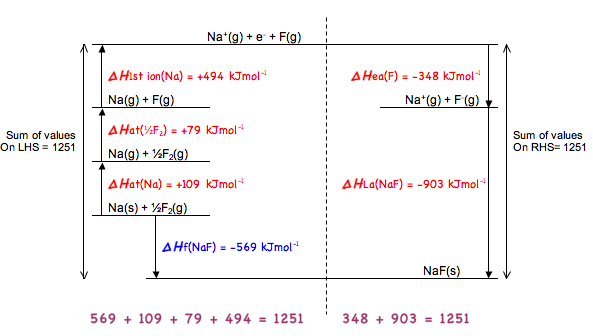
Using this method we can find out the lattice association (or lattice formation) enthalpy
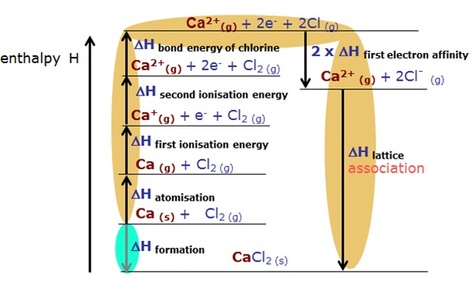
Below is what happens with a Group 2 element
Enthalpy Definitions
Lattice association/formation enthalpy -
The enthalpy change when one mole of crystalline/solid ionic substance is formed from its constituent ions in their gaseous state under standard conditions
Lattice dissociation enthalpy -
The enthalpy change when one mole of crystalline/solid ionic substance breaks up into its constituent ions in their gaseous state under standard conditions
First Ionisation Energy -
The enthalpy change when one mole of gaseous atoms loses one electron per atom under standard conditions
Second Ionisation Energy -
The enthalpy change when one mole of gaseous +1 ions lose one electron per atom under standard conditions
First Electron Affinity Enthalpy -
The Enthalpy change when one mole of gaseous atoms gains one electron per atom under standard conditions
Second Electron Affinity Enthalpy -
The Enthalpy change when one mole of gaseous -1 ions gain an electron per atom under standard conditions
Hydration Enthalpy -
The Enthalpy change when one mole of gaseous ions become hydrated or aqueous
Enthalpy of Solution -
The Enthalpy change when one mole of an ionic solid dissolves into enough water to completely separate all the ions
Enthalpy of Formation -
The Enthalpy change when one mole of a substance is formed from its constituent elements in their standard states under standard conditions
Enthalpy of Combustion -
The Enthalpy change when one mole of a substance is completely combusted in oxygen in their standard states under standard conditions
Enthalpy of Atomisation -
The Enthalpy change when one mole of gaseous atoms are produced from one mole of standard state elements
Enthalpy of Bond Dissociation -
Enthalpy change when one mole of a covalent bond is broken in the gaseous state
The enthalpy change when one mole of crystalline/solid ionic substance is formed from its constituent ions in their gaseous state under standard conditions
Lattice dissociation enthalpy -
The enthalpy change when one mole of crystalline/solid ionic substance breaks up into its constituent ions in their gaseous state under standard conditions
First Ionisation Energy -
The enthalpy change when one mole of gaseous atoms loses one electron per atom under standard conditions
Second Ionisation Energy -
The enthalpy change when one mole of gaseous +1 ions lose one electron per atom under standard conditions
First Electron Affinity Enthalpy -
The Enthalpy change when one mole of gaseous atoms gains one electron per atom under standard conditions
Second Electron Affinity Enthalpy -
The Enthalpy change when one mole of gaseous -1 ions gain an electron per atom under standard conditions
Hydration Enthalpy -
The Enthalpy change when one mole of gaseous ions become hydrated or aqueous
Enthalpy of Solution -
The Enthalpy change when one mole of an ionic solid dissolves into enough water to completely separate all the ions
Enthalpy of Formation -
The Enthalpy change when one mole of a substance is formed from its constituent elements in their standard states under standard conditions
Enthalpy of Combustion -
The Enthalpy change when one mole of a substance is completely combusted in oxygen in their standard states under standard conditions
Enthalpy of Atomisation -
The Enthalpy change when one mole of gaseous atoms are produced from one mole of standard state elements
Enthalpy of Bond Dissociation -
Enthalpy change when one mole of a covalent bond is broken in the gaseous state
Entropy
Entropy is the measure of disorder.
Gas is more disordered than a liquid which is more disordered than a solid
If there is an increases in particles then there is an increase in entropy
It is measured in JK^-1mol^-1 and the symbol S
For a reaction to occur the total entropy must be positive
To work this out the entropy change of the surroundings must also be deduced as well as the entropy of the reaction.
The Entropy change of a reaction is worked out in the same way as the enthalpy change of a reaction
Gas is more disordered than a liquid which is more disordered than a solid
If there is an increases in particles then there is an increase in entropy
It is measured in JK^-1mol^-1 and the symbol S
For a reaction to occur the total entropy must be positive
To work this out the entropy change of the surroundings must also be deduced as well as the entropy of the reaction.
The Entropy change of a reaction is worked out in the same way as the enthalpy change of a reaction
Gibbs Free-energy
For a spontaneous reaction to be feasible then Gibbs free-energy must be zero or negative
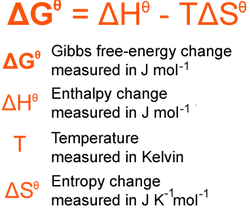
Gibbs free-energy is worked out using the below equation but beware of the units
Gibbs free-energy is worked out using the below equation but beware of the units
If you are asked to work at what temperature a reaction becomes feasible remember to make Gibbs free-energy equal zero. From there you can rearrange the equation to work out whatever you want.