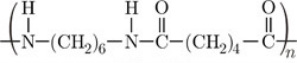Addition Polymers
Addition polymers were introduced at AS
They are created from alkenes (called monomers) in a free radical addition reaction
They don't biodegrade
They are created from alkenes (called monomers) in a free radical addition reaction
They don't biodegrade
Condensation Polymers
Condensation polymers appear in amino acids
Each monomer has at least two functional groups
They are formed from condensation reactions - loss of a water molecule
They are biodegradable as they return back to their monomers through hydrolysis
Each monomer has at least two functional groups
They are formed from condensation reactions - loss of a water molecule
They are biodegradable as they return back to their monomers through hydrolysis
Polyamides
Polyamides have the link -CONH- from the carboxyl and amino groups
Nylon 6,6
Nylon 6,6 is made from 1,6-diaminohexane and hexane-1,6-dioic acid
Nylon 6,6
Nylon 6,6 is made from 1,6-diaminohexane and hexane-1,6-dioic acid
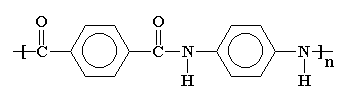
Kevlar
Kevlar is made from benzene-1,4-diamine and benzene-1,4-dioic acid
Kevlar is made from benzene-1,4-diamine and benzene-1,4-dioic acid
Polyesters
Polyesters have the link -COO- from the hydroxyl and carboxyl groups
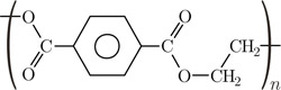
Terylene
Terylene is made from benzene-1,4-dioic acid and ethane-1,2-diol
Terylene
Terylene is made from benzene-1,4-dioic acid and ethane-1,2-diol