Everything you learnt in AS Periodicity probably will appear on this exam
Period 3 Reactions with Water
Sodium
Sodium reacts vigorously with water
Na(s) + H2O(l) → 2NaOH(aq) + H2(g)
It produces a strongly alkaline solution (pH 12-14)
Na(s) + H2O(l) → 2NaOH(aq) + H2(g)
It produces a strongly alkaline solution (pH 12-14)
Magnesium
Magnesium reacts slowly with water
Mg(s) + 2H2O(l) → Mg(OH)2(aq) + H2(g)
This produces a partially alkaline solution (pH 10) as magnesium hydroxide is only sparingly soluble
It reacts faster with steam
Mg(s) + 2H2O(g) → Mg2O(s) + H2(g)
Mg(s) + 2H2O(l) → Mg(OH)2(aq) + H2(g)
This produces a partially alkaline solution (pH 10) as magnesium hydroxide is only sparingly soluble
It reacts faster with steam
Mg(s) + 2H2O(g) → Mg2O(s) + H2(g)
Period 3 Reactions with Oxygen
Sodium
Sodium vigorously burns with a yellow flame forming white sodium oxide
2Na(s) + 1/2O2(g) → Na2O(s)
Na2O has a giant ionic structure
2Na(s) + 1/2O2(g) → Na2O(s)
Na2O has a giant ionic structure
Magnesium
Magnesium strips vigorously burn with a very bright white flame forming white magnesium oxide
2Mg(s) + O2(g) → 2MgO(s)
MgO has a giant ionic structure
2Mg(s) + O2(g) → 2MgO(s)
MgO has a giant ionic structure
Aluminium
Aluminium powder slowly burns with a white flame forming white aluminium oxide
4Al(s) + 3O2(g) → 2Al2O3(s)
Al2O3 has a giant ionic structure and won't react with water
4Al(s) + 3O2(g) → 2Al2O3(s)
Al2O3 has a giant ionic structure and won't react with water
Silicon
Silicon slowly burns
Si(s) + O2(g) → SiO2(s)
SiO2 has a macromolecular structure and won't react with water
Si(s) + O2(g) → SiO2(s)
SiO2 has a macromolecular structure and won't react with water
Phosphorus
White Phosphorus spontaneously combusts with a bright white flame
P4(s) + 5O2(g) → P4O10(s)
P4O10 has a simple molecular structure
P4(s) + 5O2(g) → P4O10(s)
P4O10 has a simple molecular structure
Sulphur
Sulphur burns steadily with a blue flame
S(s) + O2(g) → SO2(g)
SO2 has a simple molecular structure
S(s) + O2(g) → SO2(g)
SO2 has a simple molecular structure
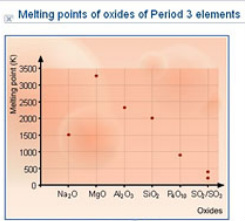
Oxide Trends
Sodium oxide has giant ionic structure - strong ionic bonds between ions need to be broken
Magnesium oxide has giant ionic structure - strong ionic bonds between ions need to be broken (stronger than sodium oxide as magnesium has a 2+ charge and so has stronger ionic bonds)
Aluminium Oxide has giant ionic structure - strong ionic bonds between ions that behave slightly covalently thus lower than MgO
Silicon Dioxide has a macromolecular structure - strong covalent bonds need to be broken
Phosphorus Oxide is simple molecular - weak van der waal's forces
Sulphur Dioxide and Trioxide are simple molecular - weak van der waal's forces (sulphur trioxide is bigger so has stronger van der waal's)
Magnesium oxide has giant ionic structure - strong ionic bonds between ions need to be broken (stronger than sodium oxide as magnesium has a 2+ charge and so has stronger ionic bonds)
Aluminium Oxide has giant ionic structure - strong ionic bonds between ions that behave slightly covalently thus lower than MgO
Silicon Dioxide has a macromolecular structure - strong covalent bonds need to be broken
Phosphorus Oxide is simple molecular - weak van der waal's forces
Sulphur Dioxide and Trioxide are simple molecular - weak van der waal's forces (sulphur trioxide is bigger so has stronger van der waal's)
Oxide Reactions with Water
Acid - Base Reactions
Sodium and Magnesium Oxide are bases
Na2O + HCl →2NaCl + H2O
MgO + H2SO4 → MgSO4 + H2O
Aluminium oxide doesn't react with water which is why aluminium sheets don't rust because aluminium oxide forms an inert protective layer
Aluminium oxide is amphoteric (alkali and acid) so it reacts with both bases and acids
Al2O3 + 3H2SO4 → Al2(SO4)3 + 3H2O
Al2O3 + 2NaOH + 3H2O → 2NaAl(OH)4
Silicon Oxide, Phosphorous Oxide and the Sulphur Oxides are acids
SiO2 + 2NaOH → Na2SiO3 + H2O
P4O10 + 12NaOH → 4Na3PO4 + 6H2O
SO2 + 2NaOH → Na2SO3 + H2O
etc.
Na2O + HCl →2NaCl + H2O
MgO + H2SO4 → MgSO4 + H2O
Aluminium oxide doesn't react with water which is why aluminium sheets don't rust because aluminium oxide forms an inert protective layer
Aluminium oxide is amphoteric (alkali and acid) so it reacts with both bases and acids
Al2O3 + 3H2SO4 → Al2(SO4)3 + 3H2O
Al2O3 + 2NaOH + 3H2O → 2NaAl(OH)4
Silicon Oxide, Phosphorous Oxide and the Sulphur Oxides are acids
SiO2 + 2NaOH → Na2SiO3 + H2O
P4O10 + 12NaOH → 4Na3PO4 + 6H2O
SO2 + 2NaOH → Na2SO3 + H2O
etc.