Kinetics
The study of the rate at which a chemical reaction occurs
You can't forget Kinetics from AS. A2 builds upon this
An increase in temperature increases the average kinetic energy of the particles hence increasing the fraction of particles with the activation energy required for a successful collision. This increases the rate of reaction
An increase in concentration and pressure increases the likelihood of a successful collision as particles are closer together. This increases the rate of reaction
A catalyst lowers the activation energy thus more particles have the activation energy, increasing the chance of a successful collision. This increases the rate of reaction
An increase in surface area increases the likelihood of a successful collision as there are more particles available to react. This increases the rate of reaction
An increase in concentration and pressure increases the likelihood of a successful collision as particles are closer together. This increases the rate of reaction
A catalyst lowers the activation energy thus more particles have the activation energy, increasing the chance of a successful collision. This increases the rate of reaction
An increase in surface area increases the likelihood of a successful collision as there are more particles available to react. This increases the rate of reaction
Reaction Rates
Chemical reactions don't occur instantaneously and so the concentration over the period of a reaction can be measured
Lets consider the reaction of
A → B
Lets consider the reaction of
A → B
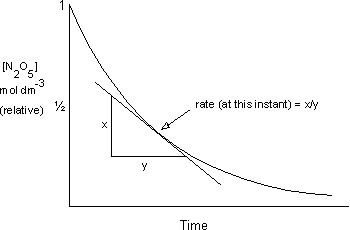
The steeper the curve the faster the rate
The initial slopes are steepest because at the beginning there is the greatest concentration of reactants
The initial slopes are steepest because at the beginning there is the greatest concentration of reactants
This means that
Change in products or reactants = Rate
Change in time
Change in products or reactants = Rate
Change in time
However, the two lines don't have a constant gradient which means a changing rate
This means that to take the rate only the gradient at one point can be taken using the tangent
This means that to take the rate only the gradient at one point can be taken using the tangent
In words the rate of reaction can be expressed as
The increase/decrease in concentration of a product/reactant as a function of time
The increase/decrease in concentration of a product/reactant as a function of time
Rate Equation
The rate equation's relation to the general equation is:
As you can see the coefficients don't appear in the equation
This means the rate equation must be found through experimentation
'k' stands for the reaction constant. It remains constant for that one reaction but it's units change
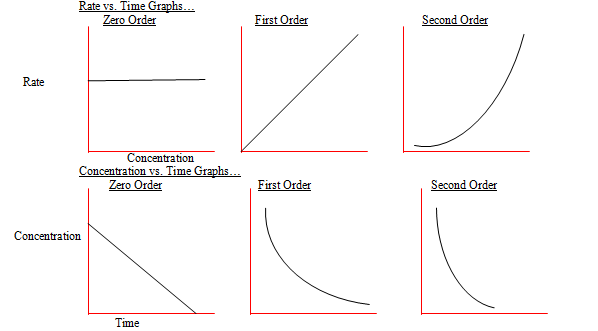
When the power is one the reaction is first order
When the power is two the reaction is second order etc.
This means the rate equation must be found through experimentation
'k' stands for the reaction constant. It remains constant for that one reaction but it's units change
When the power is one the reaction is first order
When the power is two the reaction is second order etc.
Working out the reaction constant's unit
This is an example frame
Discovering the orders in a reaction
Some of the reactants react quicker in a reaction which increases the rate of the equation
How much faster they do this by is called the order and on the reaction equation it is the power
How much faster they do this by is called the order and on the reaction equation it is the power
Rate Determining Step
The rate determining step is the intermediate reaction which determines the rate of the whole reaction
In every process there is always a point in the process which determines how fast the end result is reached.
When making a cup of coffee the rate determining step is the length of time the water takes to boil as this takes the longest of all the processes involved in the making process.
In a chemical reaction you can't time the rate determining step.
Instead you use the intermediate reactions and the rate equation
Take the equation A + D → F + G
A + B → C (1)
C + D → B + E (2)
E → F + G (3)
Rate = k[A][B]
Using this information it shows that Step 1 is the rate determining step as both chemicals in the rate equation appear in step 1.
This information also tells us that B is a catalyst and C and E are intermediates.
In every process there is always a point in the process which determines how fast the end result is reached.
When making a cup of coffee the rate determining step is the length of time the water takes to boil as this takes the longest of all the processes involved in the making process.
In a chemical reaction you can't time the rate determining step.
Instead you use the intermediate reactions and the rate equation
Take the equation A + D → F + G
A + B → C (1)
C + D → B + E (2)
E → F + G (3)
Rate = k[A][B]
Using this information it shows that Step 1 is the rate determining step as both chemicals in the rate equation appear in step 1.
This information also tells us that B is a catalyst and C and E are intermediates.
If Step 2 or 3 was the rate determining step then any chemicals from earlier reactions can appear in the rate equation as they too contribute to the time of the reaction.