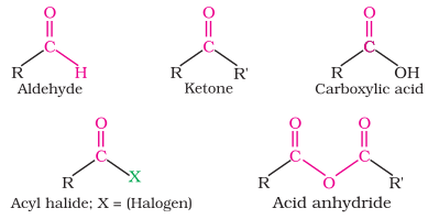
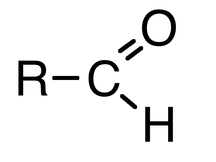
The Carbonyl Group is a C=O
Aldehydes and Ketones
AldehydesAldehydes are named with the suffix -al
The carbonyl group is at the end of the carbon chain Methanal is the simplest aldehyde Written as RCHO not like an alcohol RCOH KetonesKetones are named with the suffix -one
The carbonyl group is within the carbon chain Propanone is the simplest ketone From pentanone and onwards the group needs to be labelled - You can have propan-2-one or propan-3-one |
You still need to know everything about ketones and aldehydes from AS
Physical Properties
|
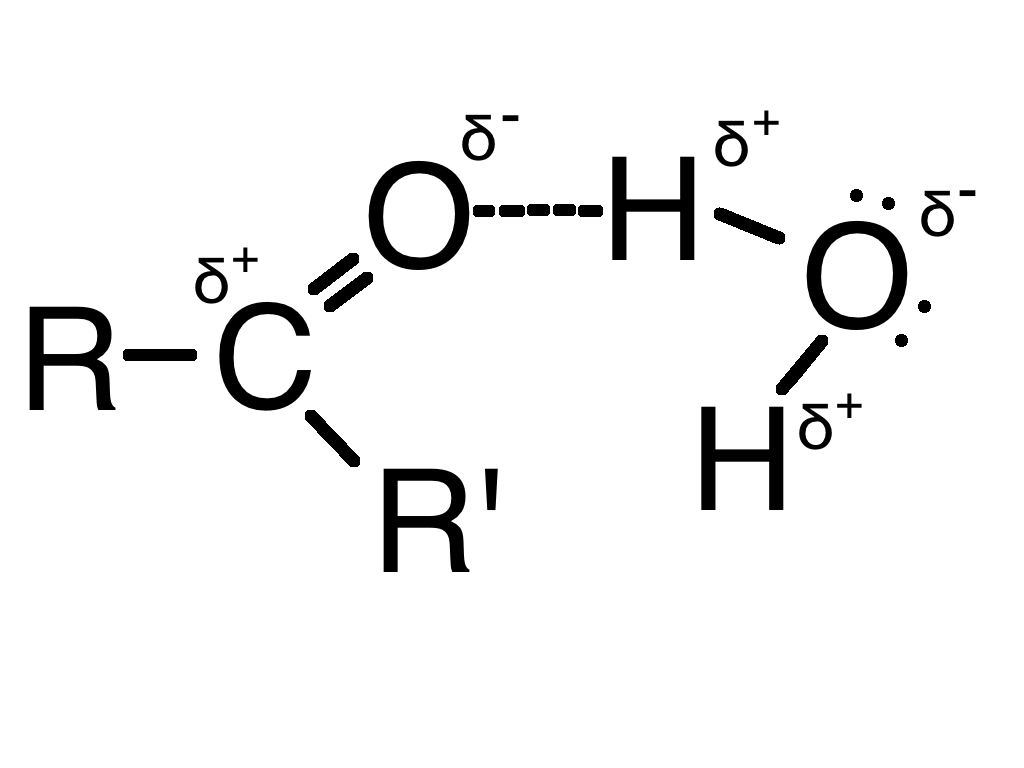
The carbonyl group is polar because oxygen is more electronegative than carbon
This means the strongest intermolecular force between two carbonyl group compounds is dipole-dipole However, they can hydrogen bond with compounds like alcohol or water This makes them soluble in water Longer chained carbonyl compounds are less soluble as the longer chain gets in the way of the hydrogen bonds The double bond makes the carbonyl group very polar This makes the carbonyl carbon very reactive with electron rich nucleophiles They react with nucleophiles using the mechanism Nucleophilic Addition |
|
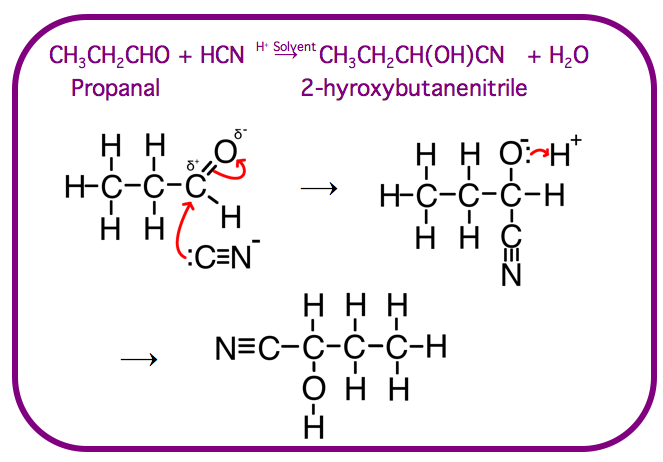
Nucleophilic Addition
Nucleophilic Addition is mechanism where a nucleophile is added to a carbonyl group in the presence of a H+ solvent
As well as the cyanide ion you need to know how nucleophilic addition occurs with the following nucleophiles:
Reduction
Aldehydes and Ketones will be reduced to their relative alcohol via nucleophilic addition
It requires the reducing agent Tetrahydroborate (NaBH4) with a H+ solvent
It requires the reducing agent Tetrahydroborate (NaBH4) with a H+ solvent
Nucleophilic Addition Elimination
Happens in acyl chlorides, carboxylic acids and anhydrides