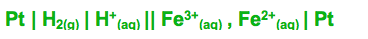Oxidation is loss of electrons and gain in oxidation state
Reduction is gain of electrons and reduction in oxidation state
Oxidation state is the value given to the ions
Oxidising Agent accepts electrons (is reduced)
Reducing Agent donates electrons (is oxidised)
Reduction is gain of electrons and reduction in oxidation state
Oxidation state is the value given to the ions
Oxidising Agent accepts electrons (is reduced)
Reducing Agent donates electrons (is oxidised)
Half-Equations
To help balance a redox equation you can write out what happens to each individual ion - this equation includes electrons.
You may need to add Water and H+ ions or OH- ions
You then combine the equations by balancing the electrons
Take the equation between the dichromate ion and the sulphur trioxide ion
You may need to add Water and H+ ions or OH- ions
You then combine the equations by balancing the electrons
Take the equation between the dichromate ion and the sulphur trioxide ion
The charges should balance on any equation you do
Electrochemistry
Electrochemical Cells
Electrochemical cells link up two half equations through a circuit enabling a current to be made.
At the positive electrode (anode) we have Zinc oxidised to Zinc (II) ions. This allows the flow of electrons to pass through the wire and be used at the negative electrode (cathode) where Copper (II) ions are reduced to copper.
The salt bridge is there to enable the circuit to be completed by letting salts form.
The salt bridge is there to enable the circuit to be completed by letting salts form.
Standard Hydrogen Electrode
The Standard Hydrogen Electrode requires:
The Platinum electrode is required because a solid is required to link up to the circuit. This is the case whenever a half-cell doesn't have a solid product.
- Hydrogen Gas
- 1 moldm^-3 H+ (or HCl) solution
- A Platinum Electrode
- 298K and 100kPa
The Platinum electrode is required because a solid is required to link up to the circuit. This is the case whenever a half-cell doesn't have a solid product.
Electrode Potential
Because a current is created by a half-cell each half cell creates its own potential difference (voltage).
This is called the Electrode Potential
The electrode potential decides which way round the overall reaction occurs
The more positive the electrode potential the stronger the oxidating power of the reactants
The more negative the stronger the reducing power of the products
This is all in relation to the standard Hydrogen Electrode potential which is 0V always
Take the Zinc/Copper cell
This is called the Electrode Potential
The electrode potential decides which way round the overall reaction occurs
The more positive the electrode potential the stronger the oxidating power of the reactants
The more negative the stronger the reducing power of the products
This is all in relation to the standard Hydrogen Electrode potential which is 0V always
Take the Zinc/Copper cell
The conventional written out method of drawing a cell is:
| stands for change in phase/state
|| stands for salt Bridge
More positive electrode potential is put on the right
When there isn't a solid formed then a platinum electrode is used and put in on the edge
Plus if there isn't a change in state a coma is used
|| stands for salt Bridge
More positive electrode potential is put on the right
When there isn't a solid formed then a platinum electrode is used and put in on the edge
Plus if there isn't a change in state a coma is used
Types of Cell
Non-Rechargeable Cells

Non-rechargeable cells are cells where the reaction can't safely be reversed inside the cell.
Zinc and Manganese Dioxide are the two chemicals used in a non-rechargeable cell
Zinc and Manganese Dioxide are the two chemicals used in a non-rechargeable cell
Rechargeable Cells

Rechargeable cells are cells where the chemical reaction can be reversed inside.
Lead and Lead(IV) dioxide are used in one type, other examples are nickel-cadmium or lithium ion cells.
Lead and Lead(IV) dioxide are used in one type, other examples are nickel-cadmium or lithium ion cells.
Advantages and Disadvantages
Cost: Non-rechargeable batteries are cheaper in the short run but rechargeable batteries don't have to be replaced
Lifetime: Non-rechargeable batteries will run for longer but rechargeable batteries don;t have t be disposed of when they run out
Power: Rechargeable batteries supply more power than non-rechargeable
Resources and Waste: Non-rechargeable batteries need to be constantly remade and chucked away however they don't contain toxic metals so aren't as hazardous when disposed of
Lifetime: Non-rechargeable batteries will run for longer but rechargeable batteries don;t have t be disposed of when they run out
Power: Rechargeable batteries supply more power than non-rechargeable
Resources and Waste: Non-rechargeable batteries need to be constantly remade and chucked away however they don't contain toxic metals so aren't as hazardous when disposed of
Fuel Cells
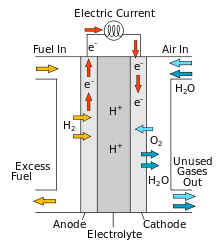
Fuel cells are cells where the chemicals are fed into the cell.
In a hydrogen-oxygen fuel cell Hydrogen and oxygen are fed in to opposite platinum electrodes. H+ ions are then filtered through to the oxygen platinum cathode where water is made.
Problems are Hydrogen needs to be created which requires electricity - process is not carbon-neutral
Hydrogen is also highly flammable.
In a hydrogen-oxygen fuel cell Hydrogen and oxygen are fed in to opposite platinum electrodes. H+ ions are then filtered through to the oxygen platinum cathode where water is made.
Problems are Hydrogen needs to be created which requires electricity - process is not carbon-neutral
Hydrogen is also highly flammable.