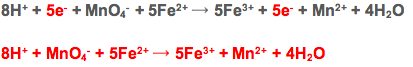A transition metal is an element that can form one or more stable ion with a partially filled d orbital
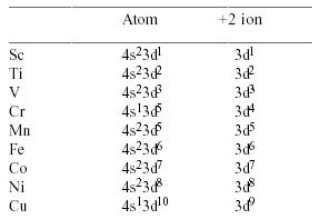
Electron Configuration
The transition metals are located in block d of the periodic table.
Their electron configuration always start with 1s2 2s2 2p6 3s2 3p6.
After that they contain the 3d and 4s groups.
Chromium and Copper differ from the next general pattern as they only have one electron in the 4s shell unlike the other metals.
After that they contain the 3d and 4s groups.
Chromium and Copper differ from the next general pattern as they only have one electron in the 4s shell unlike the other metals.
In ions the 4s shell always empties before the 3d shell because it has a lower energy level.
Redox Reactions
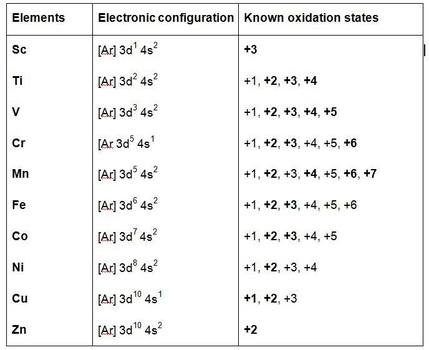
Below is a list of all the oxidation states of the transition metals in the 3d row
Only the low oxidation state ions exist by themselves.
Mn 7+ is always covalently bonded. It is commonly found as MnO4-
Mn 7+ is always covalently bonded. It is commonly found as MnO4-
Half Equations
Using half equations is very useful when balancing a reaction.
First the spectator ions are removed and the two ions taking part in the redox are separated into their own half equations
Half Equations with the Manganate (VII) ion
First the spectator ions are removed and the two ions taking part in the redox are separated into their own half equations
Half Equations with the Manganate (VII) ion
To put these two together the electrons need to balance. This means multiplying the iron (II) half equation by 5
The dichromate (VI) ion was introduced in AS when dealing with aldehydes, ketones and carboxylic acids. It works in a similar way to the manganate ion
High oxidation state metals are easily reduced in acidic conditions and Low oxidation state metals are easily oxidised in alkali conditions
High oxidation state metals are easily reduced in acidic conditions and Low oxidation state metals are easily oxidised in alkali conditions
Chromium
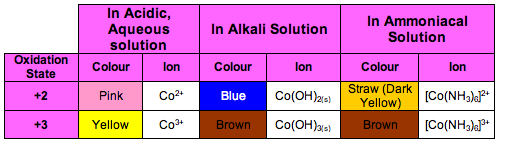
Chromium has three main oxidation states. +2, +3 and +6
You need to know what ions they form and what colour they are in acidic, aqueous and alkali conditions
You need to know what ions they form and what colour they are in acidic, aqueous and alkali conditions
Reducing Chromium with Zinc (and Iron (II))
Zinc is a very strong reducing agent and can reduce Chromium from +6 to + 3 and +2 when in acidic conditions.
This is a colour change of orange to green to blue
Iron (II) can only reduce Dichromate (VI) to Chromium (III)
This is a colour change of orange to green to blue
Iron (II) can only reduce Dichromate (VI) to Chromium (III)
Oxidising Chromium with Hydrogen Peroxide
Hydrogen Peroxide is a reducing agent in acidic solution
However, it oxidises Chromium (III) in alkaline conditions producing the chromate (VI) ion.
This colour change is green to yellow
However, it oxidises Chromium (III) in alkaline conditions producing the chromate (VI) ion.
This colour change is green to yellow
Cobalt
Cobalt has two main oxidation states of +3 and +2
You need to know what ions they form and their colours under different conditions
You need to know what ions they form and their colours under different conditions
Oxidising Cobalt with Hydrogen Peroxide
Hydrogen Peroxide oxidises Cobalt (II) to Cobalt (III) when in alkali conditions
The colour change is blue to brown
The colour change is blue to brown
Oxidising Cobalt with Air (Oxygen and water)
Cobalt (II) can be oxidised to Cobalt (III) when left to stand in air when in an ammonia solution
The colour change is yellow/straw to brown
The colour change is yellow/straw to brown
Colour and Colorimetry
Most metals and their ions are coloured.
This is because when visible light enters an ion, certain frequencies of visible light are absorbed. When this happens d orbital electrons are promoted to a higher energy level. Different wavelengths are reflected and don't have this affect giving the ion its colour.
The energy gained by the electrons can be worked out using the equation
E = hv
E is the change in energy
h is Planck's constant. It is 6.63 x 10^-34
v stands for the absorbed light's frequency
This is because when visible light enters an ion, certain frequencies of visible light are absorbed. When this happens d orbital electrons are promoted to a higher energy level. Different wavelengths are reflected and don't have this affect giving the ion its colour.
The energy gained by the electrons can be worked out using the equation
E = hv
E is the change in energy
h is Planck's constant. It is 6.63 x 10^-34
v stands for the absorbed light's frequency
Lewis Bases/Acids and Complex Ions
Bronsted-Lowry aren't the only acids and bases.
Lewis Acids and Bases are about the exchange of lone pairs of electrons. These are commonly found on nucleophiles which in this unit are refered to as ligands
Lewis Acid - Electron/lone pair donor
Lewis Base - Electron/lone pair acceptor
Ligand - Electron pair donor and molecule or ion that coordinately bonds with metals
Complex Ion - Ion containing two or more ligands (with dative/coordinate covalent bonds)
Coordination Number - Number of coordinate bonds in a complex ion
Lewis Acids and Bases are about the exchange of lone pairs of electrons. These are commonly found on nucleophiles which in this unit are refered to as ligands
Lewis Acid - Electron/lone pair donor
Lewis Base - Electron/lone pair acceptor
Ligand - Electron pair donor and molecule or ion that coordinately bonds with metals
Complex Ion - Ion containing two or more ligands (with dative/coordinate covalent bonds)
Coordination Number - Number of coordinate bonds in a complex ion
Ligands
Ligands can contain one or more lone electron pair.
Unidentate Ligand - Contains one lone electron pair
Cl-, H2O, CN-, NH3, OH- are ones you need to know
Bidentate Ligand - Contains two lone electron pairs
Unidentate Ligand - Contains one lone electron pair
Cl-, H2O, CN-, NH3, OH- are ones you need to know
Bidentate Ligand - Contains two lone electron pairs
Multidentate Ligand - Contains three or more lone electron pairs
|
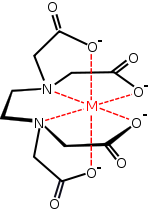
You don't necessarily need to know the structure of EDTA4-
You need to know it will coordinately bond 6 times It has an oxidation state of -4 Porphyrin coordinately bonds 4 times It has an oxidation state of 0 Porphyrin surrounding an Fe 2+ ion with the globin ligand and water ligand creates Haemoglobin |
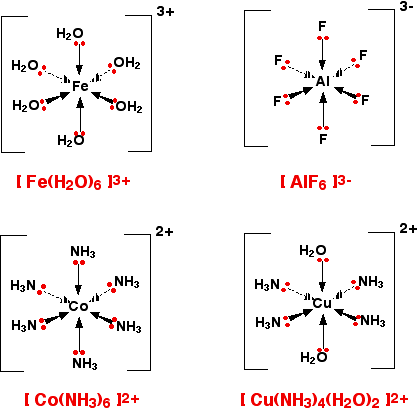
Complex Ion Shapes
You need to know the shapes and bond angles of all the ions
Most complexes contain six ligands
This creates an Octahedral shape with 90 degree bond angles
Most complexes contain six ligands
This creates an Octahedral shape with 90 degree bond angles
In [AlF6]3- the Aluminium has an oxidation state of +3 but the Fluoride ions surrounding it have an oxidation state of -1 each.
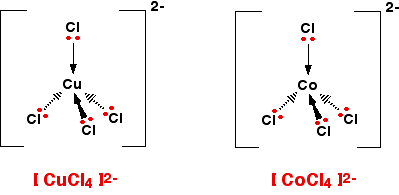
Chloride ions however are much bigger ligands. This means only four can fit round a metal.
This gives a Tetrahedral shape with a bond angle of 109.5 degrees
Chloride ions however are much bigger ligands. This means only four can fit round a metal.
This gives a Tetrahedral shape with a bond angle of 109.5 degrees
Platinum 2+ and Nickel 2+ also only bonds with four coordinate bonds
They bond with a Square planar shape and a bond angle of 90 degrees
They bond with a Square planar shape and a bond angle of 90 degrees
Silver +1 only has two coordinate bonds
It has a Linear Shape with a bond angle of 180 degrees
It has a Linear Shape with a bond angle of 180 degrees
EDTA bonds octahedrally.
Catalysts
Transition Metals are very good catalysts.
There are two types Heterogenous and Homogenous.
Heterogenous - Catalyst is in a different state/phase to the reactants
Homogenous - Catalyst is in the same state/phase as the reactants
You have:
Iron in the Haber process
Chromium (III) Oxide in the manufacture of methanol
Platinum and rhodium in catalytic converters
There are two types Heterogenous and Homogenous.
Heterogenous - Catalyst is in a different state/phase to the reactants
Homogenous - Catalyst is in the same state/phase as the reactants
You have:
Iron in the Haber process
Chromium (III) Oxide in the manufacture of methanol
Platinum and rhodium in catalytic converters
Increasing Efficiency
Increase Surface Area
Apply to an inert material. This increases the surface to mass ratio
This means that the catalyst lasts longer
They don't last forever because catalysts become 'poisoned' when impurities cover the catalyst plus they can also separate from the support medium
Apply to an inert material. This increases the surface to mass ratio
This means that the catalyst lasts longer
They don't last forever because catalysts become 'poisoned' when impurities cover the catalyst plus they can also separate from the support medium
Contact Process
Homogenous Catalyst
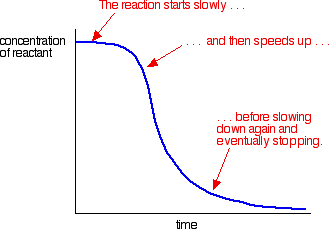
Autocatalysis
Autocatalysts are catalysts produced in the reaction they are acting as a catalyst to.
This means the reaction starts off slowly as there isn't a catalyst and then speeds up as the catalyst is gradually made
This means the reaction starts off slowly as there isn't a catalyst and then speeds up as the catalyst is gradually made