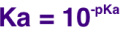Bronsted-Lowry
A Bronsted-Lowry Bases accept protons (H+ ions)
A Bronsted Lowry Acids donate protons (H+ ions)
A Bronsted Lowry Acids donate protons (H+ ions)
Strong or Weak?
The strength of a base or acid depends on the degree of dissociation (the amount of protons produced).
This means the more an acid dissociates the stronger it is.
Hyrochloric acid (HCl) "fully" dissociates into H+ ions and Cl- ions when in water. This reaction is in equilibrium but lies far to the right. HCl is considered a 'strong' acid and NaOH is considered a 'strong' base.
Carboxylic Acids (e.g. CH3COOH) slightly dissociate, in water, into carboxylate ions (CH3COO-) and H+ ions. It however is in an equilibrium that lies to the left. Carboxylic acids are considered 'weak' acids and ammonia (NH3) is considered a 'weak' base.
Water itself can be slightly acidic as it slightly dissociates into Hydroxide ions and hydroxonium ions. However the concentration of H+ and OH- is so small that the concentration of H2O is considered to be 100% of it's original value.
This means the more an acid dissociates the stronger it is.
Hyrochloric acid (HCl) "fully" dissociates into H+ ions and Cl- ions when in water. This reaction is in equilibrium but lies far to the right. HCl is considered a 'strong' acid and NaOH is considered a 'strong' base.
Carboxylic Acids (e.g. CH3COOH) slightly dissociate, in water, into carboxylate ions (CH3COO-) and H+ ions. It however is in an equilibrium that lies to the left. Carboxylic acids are considered 'weak' acids and ammonia (NH3) is considered a 'weak' base.
Water itself can be slightly acidic as it slightly dissociates into Hydroxide ions and hydroxonium ions. However the concentration of H+ and OH- is so small that the concentration of H2O is considered to be 100% of it's original value.
pH scale
|
pH is a scale to show the acidity of compounds.
It is defined as: pH = -log[H+] ([A] means the concentration of A - this is required wherever used in the exam) You don't need to worry about what this means. You just need to be able to use it. 'log' is found on every scientific calculator. It may say log10. Don't use the ln or loge button. To go backwards you use the below equation: |
|
What this shows is that an acid or base can have a range of pHs as it depends on the concentration of H+ ions. This means that
Acid Dissociation Constants
Acid Dissociation Constants are equilibrium constants with a different name
Strong Acids are considered to "fully" dissociate so the moles of acid are the same as the hydroxide ions if in a 1:1 reaction and because volume doesn't change during the reaction the concentration's are considered the same
[HCl]at start = [H+]at equilibrium
In Diprotic Acids though:
[H2SO4]at start = 2[H+]at equilibrium
However in weak acids the same rule can't be applied.
For ethanoic acid the acid dissociation constant would be just like the equilibrium constant:
Strong Acids are considered to "fully" dissociate so the moles of acid are the same as the hydroxide ions if in a 1:1 reaction and because volume doesn't change during the reaction the concentration's are considered the same
[HCl]at start = [H+]at equilibrium
In Diprotic Acids though:
[H2SO4]at start = 2[H+]at equilibrium
However in weak acids the same rule can't be applied.
For ethanoic acid the acid dissociation constant would be just like the equilibrium constant:
In this reaction the same number of moles are created each of H+ ions and CH3COO- ions. This means the equation can be written in a more helpful way when working out pH
In these equations you can assume [CH3COOH]at start = [CH3COOH]at equilibrium because so little dissociates
Water Dissociation Constant
The Water Dissociation Constant is just another acid dissociation constant.
However because water dissociates so little the [H2O] at equilibrium is considered to be the whole number and so is removed from the equation (Mathematicians just deal with this).
This gives you:
Kw = [H+][OH-]
This is used when finding out pH of bases.
Kw always equals mol^2dm^-6
However because water dissociates so little the [H2O] at equilibrium is considered to be the whole number and so is removed from the equation (Mathematicians just deal with this).
This gives you:
Kw = [H+][OH-]
This is used when finding out pH of bases.
Kw always equals mol^2dm^-6
pKa
pKa is like pH in the sense that it is defined as:
pKa = -logKa
pKa = -logKa
Putting the equations into action
Remember: Don't round any number until you get your final answer
Discovering the pH of a strong acid
Find the pH of HCl with concentration 0.163 mol/dm^3
As [HCl] = [H+] then:
pH = -log0.163 = 0.79
Always give pH to two decimal places
As [HCl] = [H+] then:
pH = -log0.163 = 0.79
Always give pH to two decimal places
Discovering the pH of a strong base
Find the pH of NaOH with concentration of 0.1 mol/dm-3 and Kw value of 1 x 10^-14
Using Kw = [H+][OH-] then
Kw = [H+]
[OH-]
1 x 10^-14 = 1 x 10^-13
0.1
pH = -log1 x 10^-13 = 13.00
Using Kw = [H+][OH-] then
Kw = [H+]
[OH-]
1 x 10^-14 = 1 x 10^-13
0.1
pH = -log1 x 10^-13 = 13.00
Discovering the pH of a weak acid
Find the pH of a 0.02 moldm^-3 solution of ethanoic acid when Ka is 1.3 x 10^-5 moldm^-3
As [H+] = [CH3COOH] we can write the equation as:
As [H+] = [CH3COOH] we can write the equation as:
Discovering the pH of a weak base
This isn't done at A2
Acid-Base Titrations (Reactions)
This is when you add acid to a base (generally an alkali - an aqueous base) until it is neutralised as shown by indicators and vice versa.
Titration Curves
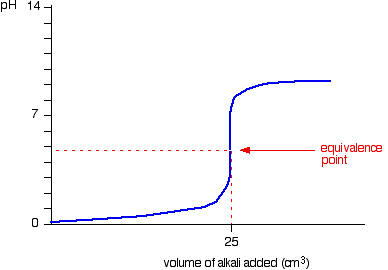
The change in pH can be plotted
Here is a reaction with a weak base being added to a strong acid. The equivalence point is the steepest point on the graph. This is where the acid or base is considered to be neutralised.
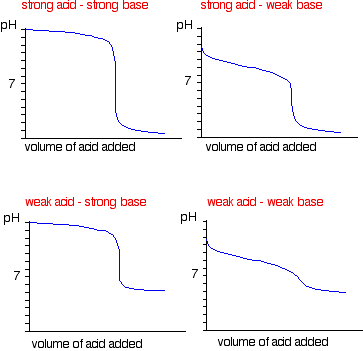
You need to know the other graphs for monoprotic acids and bases
You need to know the other graphs for monoprotic acids and bases
Indicators
To know when the equivalence point has been reached we use indicators which at a certain pH change visible characteristics.
However different indicators change at different times and are so used for different reactions
However different indicators change at different times and are so used for different reactions
The range of the indicator needs to be completely in the equivalence point (where the graph is most vertical).
This is why there isn't a suitable indicator for a reaction between a weak acid and weak base as the equivalence point is virtually non-existent.
In an exam you might given a table with indicators and their ranges.
If the question is about a weak acid to a strong base then go for the highest range there unless you know otherwise
Strong acid to weak base then go for the lowest range unless you know otherwise
Strong acid to strong base then go for the indicator with the most obvious change like phenolphlalein.
This is why there isn't a suitable indicator for a reaction between a weak acid and weak base as the equivalence point is virtually non-existent.
In an exam you might given a table with indicators and their ranges.
If the question is about a weak acid to a strong base then go for the highest range there unless you know otherwise
Strong acid to weak base then go for the lowest range unless you know otherwise
Strong acid to strong base then go for the indicator with the most obvious change like phenolphlalein.
Diprotic Acids and Bases
Some Acids and Bases donate or accept two protons like sulphuric acid.
For a diprotic acid the two protons are released in separate reactions creating two equivalence points
For a diprotic acid the two protons are released in separate reactions creating two equivalence points
Buffers
Buffers are solutions that resist changes in pH levels when small amounts of acid or alkali are added
Acidic Buffers
Acidic Buffers are weak acids and one of their salts. They have a pH < 7
CH3COOH(aq) ⇌ H+(aq) + CH3COO-(aq) only slightly dissociates
with
CH3COONa → Na+(aq) + CH3COO-(aq) fully dissociates in water
This gives you a solution with lots of CH3COOH and CH3COO-
CH3COOH(aq) ⇌ H+(aq) + CH3COO-(aq) only slightly dissociates
with
CH3COONa → Na+(aq) + CH3COO-(aq) fully dissociates in water
This gives you a solution with lots of CH3COOH and CH3COO-
Add alkali
When an alkali is added the H+ ions from the weak acid react with the OH- ions from the alkali.
This pushes the equilibrium to the right so to keep the concentration of H+ constant and thus the pH
This pushes the equilibrium to the right so to keep the concentration of H+ constant and thus the pH
Add acid
When an acid is added the H+ concentration increases pushing the equilibrium to the left.
This makes the excess H+ ions react with the carboxylate ions keeping the H+ ion concentration constant
This makes the excess H+ ions react with the carboxylate ions keeping the H+ ion concentration constant
Basic Buffers
Basic Buffers are made from a weak base and it's salt. They have a pH > 7
NH3(aq) + H2O(l) ⇌ NH4+(aq) + OH-(aq) only slightly dissociates
with
NH4Cl(aq) → Cl-(aq) + NH4+(aq) fully dissociates in water
This creates a solution with lots of NH3 and lots of NH4+
NH3(aq) + H2O(l) ⇌ NH4+(aq) + OH-(aq) only slightly dissociates
with
NH4Cl(aq) → Cl-(aq) + NH4+(aq) fully dissociates in water
This creates a solution with lots of NH3 and lots of NH4+
Add alkali
When an alkali is added the NH4+ ions will react with the OH- ions from the alkali
This pushes the equilibrium to the left keeping the concentration of OH- constant and thus the same with H+
This pushes the equilibrium to the left keeping the concentration of OH- constant and thus the same with H+
Add Acid
When an acid is added it is dealt with in two ways.
The H+ ions either:
Both of these reactions remove the extra H+ ions keeping the pH constant
The H+ ions either:
- react with OH- ions creating water pushing equilibrium to the right so to replace the missing OH- ions
- react with NH3 creating NH4+
Both of these reactions remove the extra H+ ions keeping the pH constant
Why are Buffers Useful?
Buffers are used in many places
- Shampoos contain buffers so to counteract the alkaline soap which wouldn't make the hair shiny
- Biological washing powders contain enzymes which need a constant pH level so they can work
- Biological systems keeping things like human blood at a pH near 7.4
Working out the pH of a Buffer Solution
Buffer solution contains 0.4 moldm^-3 of HCOOH and 0.6 moldm^-3 of HCOONa with a Ka of 1.6 x 10^-4 moldm^-3
Ka = [H+][HCOO-]
[HCOOH]
The HCOO- comes from the salt
To find [H+] you need to rearrange
Ka[HCOOH] = [H+]
[HCOO-]
Because the units cancel out if you only had mole values this equation is still possible to use
Ka x HCOOH = [H+]
HCOO-
Using this [H+] = 1.07 x 10^-4 moldm^-3
pH = -log1.07 x 10^-4 = 3.97
Remember in Buffers [H+] ≠ [A-] so don't use simplified Ka equation
Ka = [H+][HCOO-]
[HCOOH]
The HCOO- comes from the salt
To find [H+] you need to rearrange
Ka[HCOOH] = [H+]
[HCOO-]
Because the units cancel out if you only had mole values this equation is still possible to use
Ka x HCOOH = [H+]
HCOO-
Using this [H+] = 1.07 x 10^-4 moldm^-3
pH = -log1.07 x 10^-4 = 3.97
Remember in Buffers [H+] ≠ [A-] so don't use simplified Ka equation
Working out pH of a Buffer Solution During a Titration
Calculate the buffer formed when 500 cm^3 of 0.4 moldm^-3 of NaOH is added to 500 cm^3 of 1 moldm^-3 of HA.
Ka = 6.25 x 10^-5
First we need to discover the moles using (vol x conc)/1000 = mol
Moles of HA = 0.5 mol
Moles of NaOH = 0.2 mol
We need to know how much of HA we have and of A-
Because HA is in excess we need to deduce the amount of NaOH from HA and add to the A- the same amount due to the equation:
HA + NaOH → H2O + NaA
Initial: 0.5 0.2 0 0
Final: 0.3 0 0.2 0.2
This means we have a buffer solution of HA 0.3 mol and A- 0.2 mol
(If you want to find out the concentrations of the buffer solution - remember that the solution is now 1000 cm^3 in volume)
We don't need the concentrations to discover the pH though
Ka x HA = [H+]
A-
[H+] = 9.375 x 10^-5
pH = 4.03
Ka = 6.25 x 10^-5
First we need to discover the moles using (vol x conc)/1000 = mol
Moles of HA = 0.5 mol
Moles of NaOH = 0.2 mol
We need to know how much of HA we have and of A-
Because HA is in excess we need to deduce the amount of NaOH from HA and add to the A- the same amount due to the equation:
HA + NaOH → H2O + NaA
Initial: 0.5 0.2 0 0
Final: 0.3 0 0.2 0.2
This means we have a buffer solution of HA 0.3 mol and A- 0.2 mol
(If you want to find out the concentrations of the buffer solution - remember that the solution is now 1000 cm^3 in volume)
We don't need the concentrations to discover the pH though
Ka x HA = [H+]
A-
[H+] = 9.375 x 10^-5
pH = 4.03