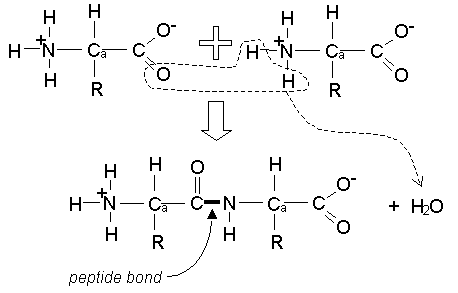Amino and Carboxyl groups together at last
Amino acids have optical isomers as they contain a chiral carbon (ignoring glycine)
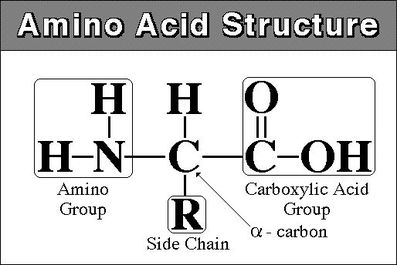
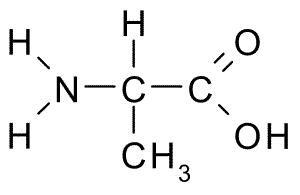
To be an amino acid the amino and carboxyl group need to be bonded to the same carbon
When naming find the longest chain and the amino group is named like the other alkyl groups
other branches-2-amino(longest chain)oic acid
To be an amino acid the amino and carboxyl group need to be bonded to the same carbon
When naming find the longest chain and the amino group is named like the other alkyl groups
other branches-2-amino(longest chain)oic acid
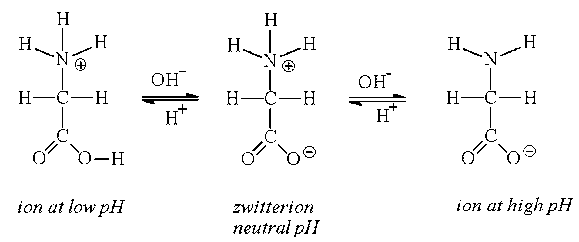
Zwitterions
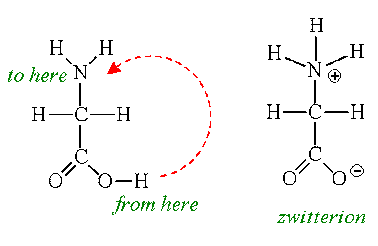
Amino acids can exist as zwitterions
Zwitterions are dipolar ions - they contain both positive and negative charge
The amino group becomes protonates and the carboxyl group becomes deprotonated
Zwitterions are dipolar ions - they contain both positive and negative charge
The amino group becomes protonates and the carboxyl group becomes deprotonated
Zwitterions become polar when in an acid or alkali
The affect here is carried through all amino and carboxyl groups
Condensation Polymers
Amino acids make condensation polymers joined together by a peptide link
Two amino acids joined together make a dipeptide
They are joined through condensation and broken through hydrolysis
Two amino acids joined together make a dipeptide
They are joined through condensation and broken through hydrolysis
If the amino acids joined together are different then you can get two possible dipeptides
These condensation polymers are proteins
These condensation polymers are proteins
Proteins
Proteins are kept in shape by the hydrogen bonds by the two polar groups NH2 and OH
The shape is vital to how the proteins work.
Because we know that amino acids can exist as zwitterions a change in pH breaks these hydrogen bonds and thus prevents the protein from doing it's function.
The shape is vital to how the proteins work.
Because we know that amino acids can exist as zwitterions a change in pH breaks these hydrogen bonds and thus prevents the protein from doing it's function.