Nucleophiles and Bases
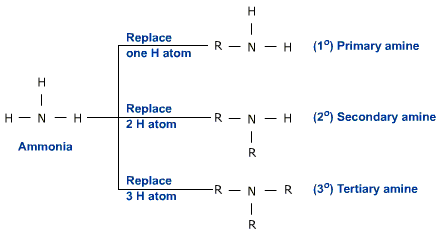
Amines are derivatives of Ammonia (NH3).
They are formed when ammonia undergoes nucleophilic substitution (from AS) with a haloalkane.
This is because they contain a lone electron pair.
During substitution with the right conditions the amines won't stop substituting as there is still a lone pair giving us primary, secondary and tertiary amines.
However, after that we get a quaternary ammonium salt which is where the reaction stops.
They are formed when ammonia undergoes nucleophilic substitution (from AS) with a haloalkane.
This is because they contain a lone electron pair.
During substitution with the right conditions the amines won't stop substituting as there is still a lone pair giving us primary, secondary and tertiary amines.
However, after that we get a quaternary ammonium salt which is where the reaction stops.
If you only want a primary amine you want an excess of ammonia. As the amount of times the amine can react is limited because the haloalkanes won't be available as other ammonias have already reacted.
If you want a tertiary amine or quaternary ammonium salt you want an excess of haloalkanes as there are more times the amine group can substitute.
If you want a tertiary amine or quaternary ammonium salt you want an excess of haloalkanes as there are more times the amine group can substitute.
Bases
They are also Bases as they can accept a proton (H+ ion).
Ammonium Salts can also be created through the amines reactions as a base.
In its reaction with HCl the H+ from HCl reacts with the lone pair of electrons creating a dative covalent bond.
The Cl- then ionically bonds with the positive ammonium.
Ammonium Salts can also be created through the amines reactions as a base.
In its reaction with HCl the H+ from HCl reacts with the lone pair of electrons creating a dative covalent bond.
The Cl- then ionically bonds with the positive ammonium.
If a strong base is then added the original amine is produced with water and a salt
Base Strengths
The strength of a base is it's ability to accept protons.
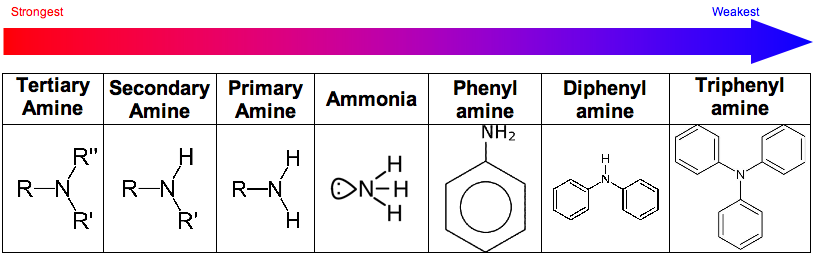
The strengths of amines varies due to electron density induction
The strengths of amines varies due to electron density induction
In tertiary amines the nitrogen has three alkyl groups inducing electron density towards the nitrogen.
This pushes the lone pair away from the nitrogen making it more available to bond with a proton.
In amines that contain the phenyl group, a different effect occurs. Instead of being pushed away the lone pair of electrons are pulled into the ring of electrons of the benzene. This means the electrons are less available to react with.
This is why electrophiles are more likely to take part in electrophilic substitution than electrophilic addition in phenylamines.
This pushes the lone pair away from the nitrogen making it more available to bond with a proton.
In amines that contain the phenyl group, a different effect occurs. Instead of being pushed away the lone pair of electrons are pulled into the ring of electrons of the benzene. This means the electrons are less available to react with.
This is why electrophiles are more likely to take part in electrophilic substitution than electrophilic addition in phenylamines.
Reducing a Nitrile
The reduction of a nitrile creates a primary amine. The catalyst is LiAlH4 with a dilute acid.
However LiAlH4 is expensive.
In industry catalytic hydrogenation is the process used with either a Nickel or Platinum catalyst and high pressure and temperature
RCN + H2 → RCH2NH2
However LiAlH4 is expensive.
In industry catalytic hydrogenation is the process used with either a Nickel or Platinum catalyst and high pressure and temperature
RCN + H2 → RCH2NH2